 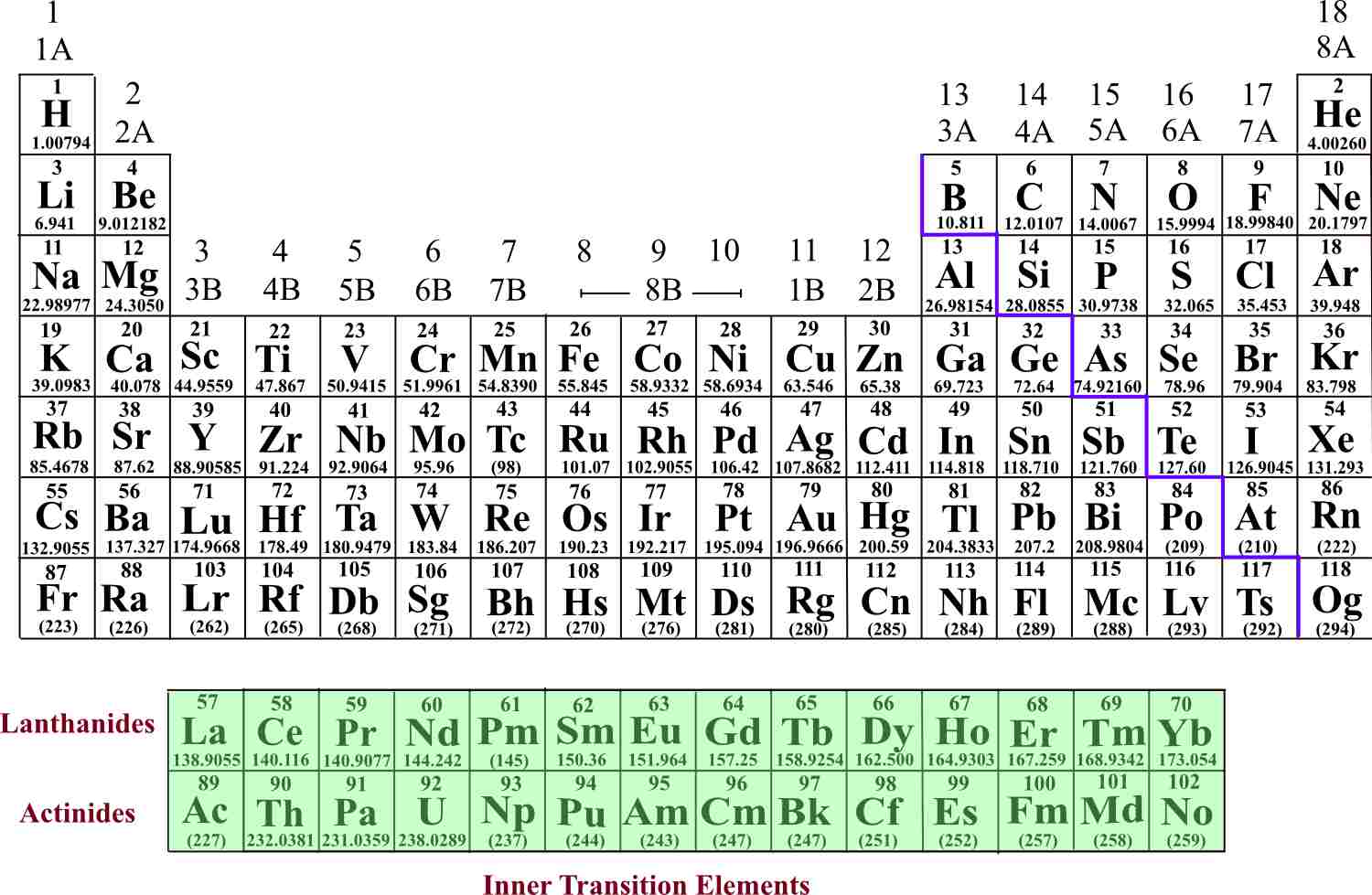
All elements in the same period have the same number of electron shells but have different numbers of electrons and protons. In each period, the elements’ atomic numbers increase from left to right. The periodic table contains seven periods 5 (nine if you count the lanthanides and actinide series). The horizontal rows across the periodic table are called periods. Because atoms naturally occur with different numbers of neutrons-known as isotopes-the atomic mass is an average of all weights of all isotopes for a given atom. Under the element symbol is the atomic weight, which is the average weight of the protons and neutrons in an atom. 4 The atomic number, located at the top left of the element symbol, signifies the number of protons in an atom’s nucleus. How elements are organized on the periodic table:Įlements are organized in horizontal rows by increasing atomic number. 2 Oxygen (O) is the most common element in the Earth’s crust. Today, the 118 chemical elements identified on the periodic table include the materials that make up all known objects in the universe.ĭid you know? The most abundant element (by mass) making up Earth3 is Iron (Fe). He anticipated others would one day be discovered, so he left open spaces in his table for additions. Mendeleev’s periodic table included 63 elements. This structure helped Mendeleev and other scientists identify similarities and differences among elements to help predict future chemical reactions. 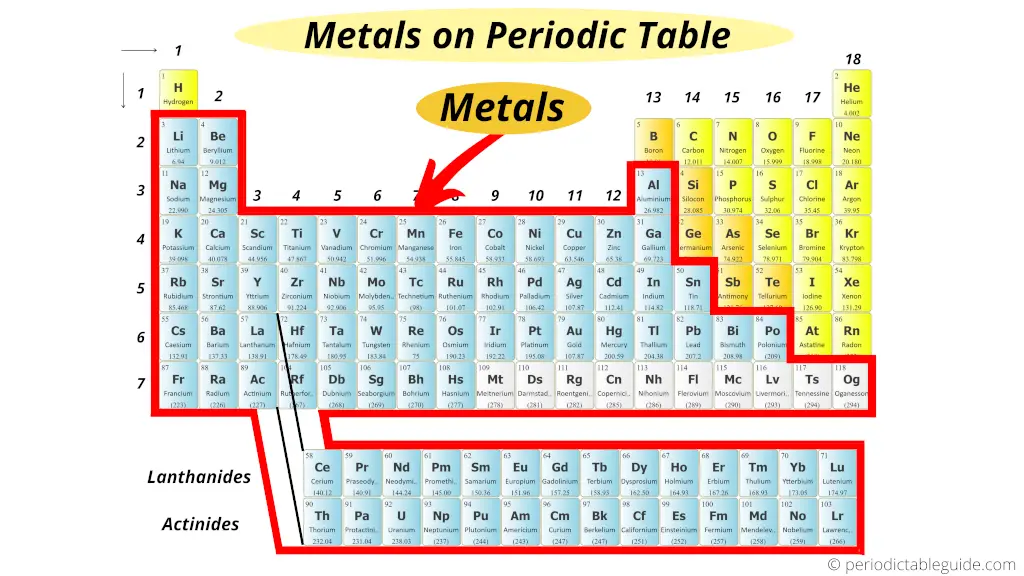
He found a pattern, listing elements by their increasing atomic number and arranging them in a chart-creating the first periodic table. In 1869, Russian chemist Dimitri Mendeleev wanted to see if there was a pattern to the chemical properties of the elements he knew. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
